The Forgotten Organ That Predicts How Long You'll Live
A new study reveals thymus health as a powerful predictor of lifespan, tied to immune function, inflammation, and chronic disease risk.
What to know
The thymus gland is a small organ located behind the sternum that plays a central role in the immune system.
New research links strong thymic health in adulthood to a lower risk of death from major diseases, including cardiovascular, metabolic, and cancer.
Poor thymic function is associated with chronic inflammation, or “inflammaging,” a key driver of aging and disease.
Lifestyle factors such as physical activity, metabolic health, and smoking status can influence the rate of thymus decline.
Targeting mitochondrial health may help support immune function and healthy aging.
There's a small gland tucked behind your sternum, about where a tie knot sits, that most physicians don't even think about after childhood. The thymus gland plays a key role in your immune system, but its job of producing T-cells is mostly done by the time you’ve reached puberty.
However, a growing body of scientific evidence suggests that this way of thinking may be outdated, and that the health of the thymus gland into adulthood might be worth considering. And now, a new paper published in Nature suggests that how well your thymus is functioning right now - whether in middle age, in your 60s, or your 70s - can predict with remarkable precision whether you will die from cancer, heart disease, metabolic disease, or pulmonary disease in the next 12 years.[1] This is independent of age, sex, smoking history, and BMI.
Let’s break down what this study from a team of scientists from Harvard, Aarhus, and UCL just showed about thymic health, and how these findings represent a paradigm shift for the field of medicine.
What Does the Thymus Gland Do?
The classical view was simple: the thymus does its important work in childhood, building your T-cell repertoire and then gradually burns itself out. The immune system, so the thinking went, would then maintain itself on the T-cells already in circulation. By adulthood, the gland is mostly replaced by fat, through a process called thymic involution, and we assumed it became largely nonfunctional.
That assumption turns out to be wrong or at least profoundly incomplete.
Science has now shown that the thymus continues producing naive T-cells throughout adult life.[2] And the rate at which it does so, and how much functional tissue remains versus how much has been replaced by fat, varies enormously between individuals. It's that variation, it turns out, that predicts who ages well and who doesn't.
What Does This New Study Show?
The team built a deep learning AI system that they trained to read routine chest CT scans and provide a thymic health score. This score quantifies how much functional tissue remains versus its fatty replacement. They applied this to 27,612 people from two large cohorts: the National Lung Screening Trial (25,031 people, 12-year follow-up) and the Framingham Heart Study (2,581 people).
The participants were “categorized as low, average or high thymic health based on the bottom 25%, middle 50% and top 25% of the population,[3]” according to how much thymic functionality was preserved.
The team then followed these two cohorts for 12 years, tracking who developed certain diseases and who died.
The findings across every major cause of death are worth sitting with. Those with high thymic health had:
- 50% lower risk of all-cause mortality over 12 years
- 36% lower likelihood of developing lung cancer
- 75% lower cardiovascular mortality across both independent cohorts
- 68% lower mortality from metabolic diseases, including diabetes
- 61% lower mortality from pulmonary disease
- 37% lower cancer progression in immunotherapy patients (data from a companion paper in the same issue of Nature)
Thymic health was predictive across virtually every major cause of death, suggesting that the immune system's role in long-term health is far more foundational than we've credited in mainstream medicine.
The Mechanism: Inflammaging and Mitochondrial Dysfunction
The researchers didn't just describe the association, they went looking for the biological clue. They measured 68 inflammation-related proteins in blood samples from 317 Framingham participants. People with worse thymic health showed elevated levels of inflammatory markers: IL-6, IL-18, VEGFA, and several pro-inflammatory chemokines. This is the molecular signature of inflammaging: the chronic, low-grade systemic inflammation that underlies most age-related disease.
They also tracked C-reactive protein (CRP) in over 1,150 people across 5–10 years. Chronically elevated CRP (≥3 mg/L) was independently associated with lower thymic health — not explained by age, sex, or smoking.
The picture that emerges is a vicious cycle: chronic inflammation accelerates thymic involution, fewer naive T-cells are produced, immune surveillance weakens, and inflammation climbs further. And sitting at the center of this cycle, increasingly, is mitochondrial dysfunction within immune cells themselves.
Mitochondrial Dysfunction and An Aging Immune System
Mitochondrial dysfunction is recognized as a central hallmark of aging. These organelles aren’t just passive “power plants”—they regulate energy production, redox balance, and even inflammatory signaling. Immune cells are especially energy-hungry.
With age, mitochondria become less efficient. They produce less ATP, generate more reactive oxygen species, and accumulate damage to their DNA and membranes. In immune cells, mitochondrial dysfunction is associated with reduced functional adaptability and a shift toward chronic, low-grade inflammatory signaling that contributes to immunosenescence and inflammaging.[4]
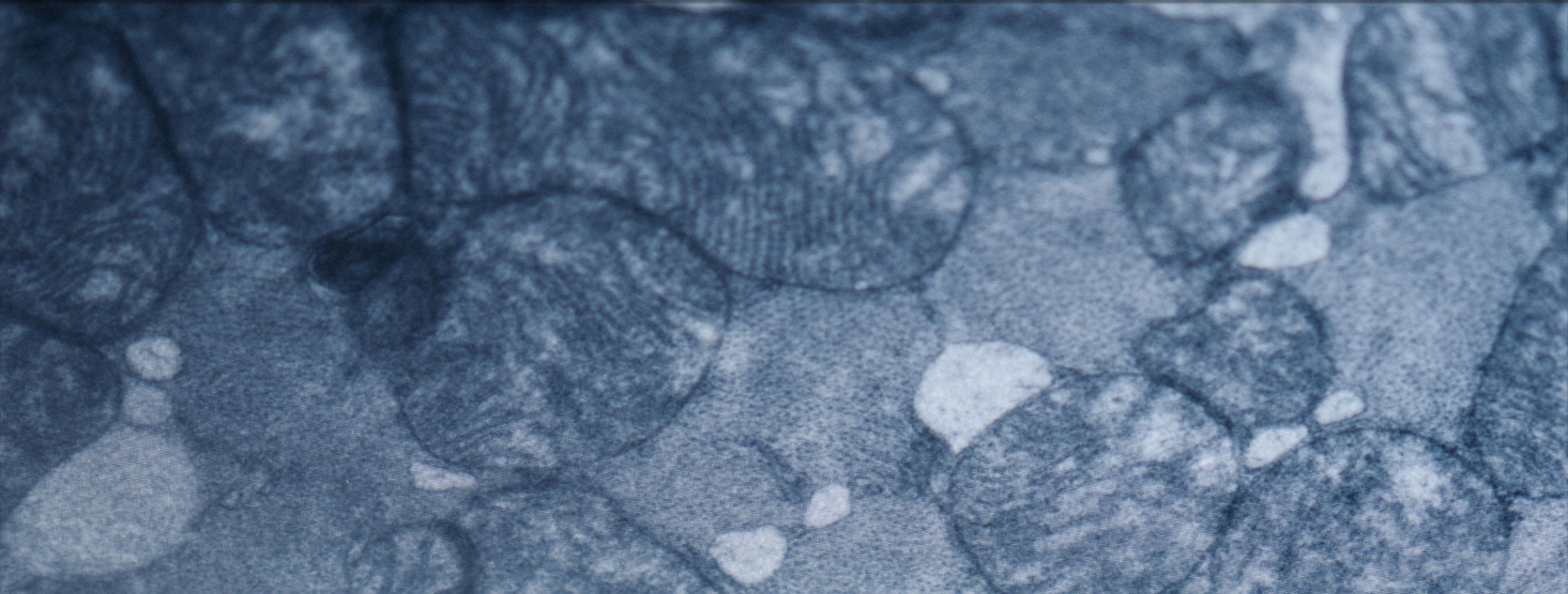
What You Can Do About It — The Mitochondria Connection
The question that immediately follows this breakthrough study is: Can we act on this? Is thymic health modifiable, or are we just measuring our fate?
The study itself establishes that thymic health is not fixed. Smoking, obesity, metabolic syndrome, and chronic inflammation all accelerate decline. Physical activity and good metabolic fitness (high HDL, healthy triglycerides) are associated with better thymic health. So lifestyle factors matter significantly.
But now, a more targeted cellular-level intervention is worth understanding. Novel molecules that target mitochondrial health are currently being studied across various aging systems, including immune health.
Companion Paper Worth Noting
A second paper in the same Nature[5] issue analyzed 1,200+ cancer patients receiving cancer immunotherapy. Those with higher thymic health had 37% lower cancer progression risk and 44% lower risk of death, even after accounting for tumor type and treatment protocol. The thymus may be one of the key determinants of who responds to checkpoint inhibitor therapy.
Path Forward
The thymus is not a relic of childhood immunity. It is an active, modifiable organ whose functional status is one of the most powerful predictors of how long and how well you'll live. A paper of this scale, in this journal, with this consistency of effect across disease categories, should shift how we think about immune health in the context of longevity medicine.
The good news is that this isn't purely observational. Lifestyle factors move the needle. And the emerging clinical evidence around mitochondrial health in immune cells offers a biologically coherent, clinically actionable path forward.
Authors

Written by
Chief Medical Officer
References
- ↑
Bernatz, S., Prudente, V., Pai, S. et al. Thymic health consequences in adults. Nature (2026). https://doi.org/10.1038/s41586-026-10242-y (https://www.google.com/url?q=https://doi.org/10.1038/s41586-026-10242-y&sa=D&source=docs&ust=1775762037141340&usg=AOvVaw2wnykeLjaLgsqFu6HdmQ1V)
- ↑
Middelkamp, V., Kekäläinen, E. Measuring thymic output across the human lifespan: a critical challenge in laboratory medicine. GeroScience 47, 6797–6806 (2025). https://doi.org/10.1007/s11357-025-01555-3 (https://www.google.com/url?q=https://doi.org/10.1007/s11357-025-01555-3&sa=D&source=docs&ust=1775762037135272&usg=AOvVaw19VhcemeZE_yTSvPmNRNte)
- ↑
Bernatz, S., Prudente, V., Pai, S. et al. Thymic health consequences in adults. Nature (2026). https://doi.org/10.1038/s41586-026-10242-y (https://www.google.com/url?q=https://doi.org/10.1038/s41586-026-10242-y&sa=D&source=docs&ust=1775762037135985&usg=AOvVaw1eMg1CrRboHZc12w4OoUHT)
- ↑
Gabriela Desdín-Micó et al. ,T cells with dysfunctional mitochondria induce multimorbidity and premature senescence.Science368,1371-1376(2020).DOI:10.1126/science.aax0860 (https://doi.org/10.1126/science.aax0860)
- ↑
Aerts et al. Thymic health as a predictor of immunotherapy outcomes. Nature (2026). doi: 10.1038/s41586-026-10243-x